

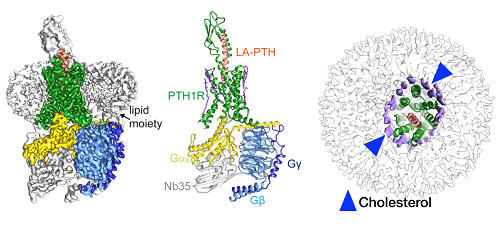
Cryo-EM structure of the human parathyroid hormone receptor-1 signaling complex. (Left, cryo-EM map; middle, structure of the complex represented by ribbon model; right, high resolution cryo-EM map revealed an ordered annular lipid belt wrapping the periphery of the receptor TMD. Green, human parathyroid hormone receptor-1 (PTH1R); orange, long-acting parathyroid hormone (LA-PTH); yellow, Ga; cyan, Gβ; blue, Gy; grey, Nb35)
Scientists at the Shanghai Institute of Materia Medica, Chinese Academy of Sciences, led by Xu Huaqiang (also known as Huaqiang Eric Xu) and Wang Mingwei, in collaboration with their counterparts at Zhejiang University School of Basic Medical Sciences led by Zhang Yan and the University of Pittsburgh School of Medicine (USA) led by Jean-Pierre Vilardaga, revealed a near-atomic resolution cryo-electron microscopy (cryo-EM) structure (3.0A) of the human parathyroid hormone type 1 receptor (PTH1R) bound to a long-acting parathyroid hormone (PTH) analog and the stimulatory G protein.
The finding was published on April 12, 2019 in Science titled “Structure and dynamics of the active human parathyroid hormone receptor-1”. It provides valuable insights into the structural basis and dynamics of PTH binding and long-term activation of the receptor, thereby laying a solid foundation for discovering novel therapeutics against osteoporosis, hypoparathyroidism, cachexia and other diseases.
PTH is a classic endocrine hormone identified over 80 years ago which plays critical and distinct roles in skeletal development, calcium homeostasis, and bone turnover. The functions of PTH are mediated primarily through binding and activation of PTH1R, a member of the class B G protein-coupled receptor (GPCR) subfamily. As a well-recognized drug target for osteoporosis, PTH1R is highly expressed in bone and kidney cells where it exerts regulatory action in calcium and phosphorus metabolism. Analogs of PTH (such as Teriparatide acetate) are presently used in the clinic to treat osteoporosis.
Previous studies have speculated that endogenous ligand binds and activates class B GPCR through a “two-step” model: the carboxyl terminal of the ligand first binds the extracellular domain (ECD) of the receptor, and then, its amino terminus inserts into the hydrophobic pocket of the transmembrane domain (TMD).
However, how the ligand dissociates from the receptor remains unknown. Parathyroid hormone not only activates its cognate receptor quickly but also dissociates rapidly. Scientists prolonged its residence time on the receptor using a long-acting agonist and subsequently captured the process of ligand dissociation by means of delicate and meticulous three-dimensional classification during imaging analysis.
The flexible ECD of the receptor remains its inherent dynamic characteristic upon ligand binding and exerts two effects on the helical ligand during the continuous movement: (1) it approaches the ligand to produce stress that prompts the ligand unwinding; and (2) it moves away from the ligand to weaken their interaction. The combination of the two leads to initial dissociation of the carboxyl end of the ligand.
This study enhances our understanding of the molecular recognition mechanism of class B GPCRs.
Coordinated by Drs. Xu and Wang, each with different but defined tasks, scientists and graduate students of the above-mentioned four research teams worked diligently and cooperated closely with each other. They overcame major technical hurdles such as low PTH1R expression levels, protein instability and difficulties in receptor-Gs complex formation, and eventually determined the three-dimensional structure of a long-acting ligand (LA-PTH) bound to PTH1R-Gs complex.
This structure demonstrates detailed interaction between LA-PTH-bound PTH1R ECD TMD and offers a comprehensive understanding of how PTH1R interacts with a peptide agonist and couples to Gs. It is the first full-length PTH1R structure in an active state and the first three-dimensional GPCR structure under long-term activation.
It also reveals the intracellular cyclic adenosine monophosphate (cAMP) signaling mechanism of PTH1R in a prolonged active state. Due to the high resolution (3.0A) of this complex, the scientists unexpectedly discovered the extensively ordered lipid distribution around the TMD which may increase conformational stability of the receptor.
This joint research “campaign” was organized by four team leaders with complementary expertise, and all of them have long been involved in structural and functional studies of class B GPCRs. Dr. Xu resolved the structure of the peptide-bound PTH1R ECD; Dr. Wang co-led the determination of crystal structures of TMD and full-length glucagon and glucagon-like peptide-1 receptors; Dr. Zhang specializes in macromolecule structural analysis using cryo-EM techniques; and Dr. Vilardaga is a renowned expert in PTH. With the spirit of collaboration and resource-sharing, Chinese and American scientists maximized each other’s strengths and took only a little over a year to conquer this global-scale scientific challenge, providing the best structural resolution among all class B GPCRs.
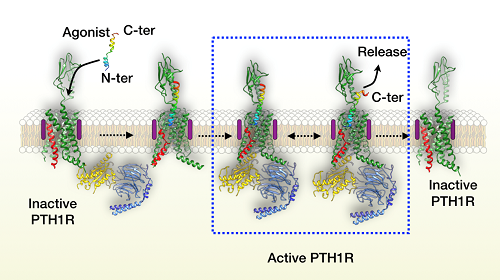
Proposed model of the human parathyroid hormone receptor-1 activation. Human parathyroid hormone receptor-1 (PTH1R) in green with transmembrane domain 6 (TM6) in red. Agonist is colored by rainbow, N- to C-terminal (N-ter and C-ter, respectively), blue to red. Peptide agonist binds to PTH1R and induces the outwards movement of TM6. GDP-bound Giheterotrimer engages the receptor. Compared with the transmembrane domain (TMD) of the receptor, extracellular domain (ECD) is wobbling around the agonist helix, leading to the separation of the peptide C-terminus from ECD and the unwinding of the helix in the C-terminal half of peptide. Subsequently, the agonist fully dissociates from the receptor.
Dr. Zhao Lihua and Ma Shanshan (PhD candidate) from the Shanghai Institute of Materia Medica, Dr. Shen Dandan from Zhejiang University and Dr. Ieva Sutkeviciute from the University of Pittsburgh are the four co-first authors of this paper. Contributing institutions also include the Van Andel Research Institute (U.S.A.), Fudan University and Harvard Medical School (U.S.A.).
The work was funded by the Chinese Academy of Sciences, the National Natural Science Foundation of China, the Ministry of Science and Technology of China, the National Health Commission of China, the National Institutes of Health (NIH, U.S.A.), the Shanghai Municipal Commission of Science and Technology, the Fudan-SIMM Joint Research Fund, the Novo Nordisk-CAS Research Fund and several career development grants from China and the United States (such as the Youth Innovation Promotion Association of the Chinese Academy of Sciences).
The paper link: https://science.sciencemag.org/content/364/6436/148
For more information, please contact:
Dr. Xu Huaqiang
E-mail: eric.xu@simm.ac.cn
Dr. Wang Mingwei
E-mail:mwwang@simm.ac.cn
Institute of Materia Medica, Chinese Academy of Sciences
(Credit: Wang Mingwei;Editor:Pan Peihua)
Source: Institute of Materia Medica,CAS